Pharma Out Licensing Challenges and How to Overcome Them
Out licensing in pharma and biotech is frequently described as a value-inflection event — a moment when an asset’s latent potential is recognised, priced, and shared with a partner capable of taking it further. That description is accurate, but incomplete. It omits what lies between the decision to out license and the moment contracts are signed: a gauntlet of process friction, organisational misalignment, and execution gaps that quietly destroy deal value or derail transactions entirely.
The out licensing process typically spans 8 to 15 months and follows four discrete stages: preparation, marketing the opportunity, due diligence, and closing. Each stage introduces its own category of risk. This piece examines the most consequential failure modes across that journey — and the strategies that resolve them.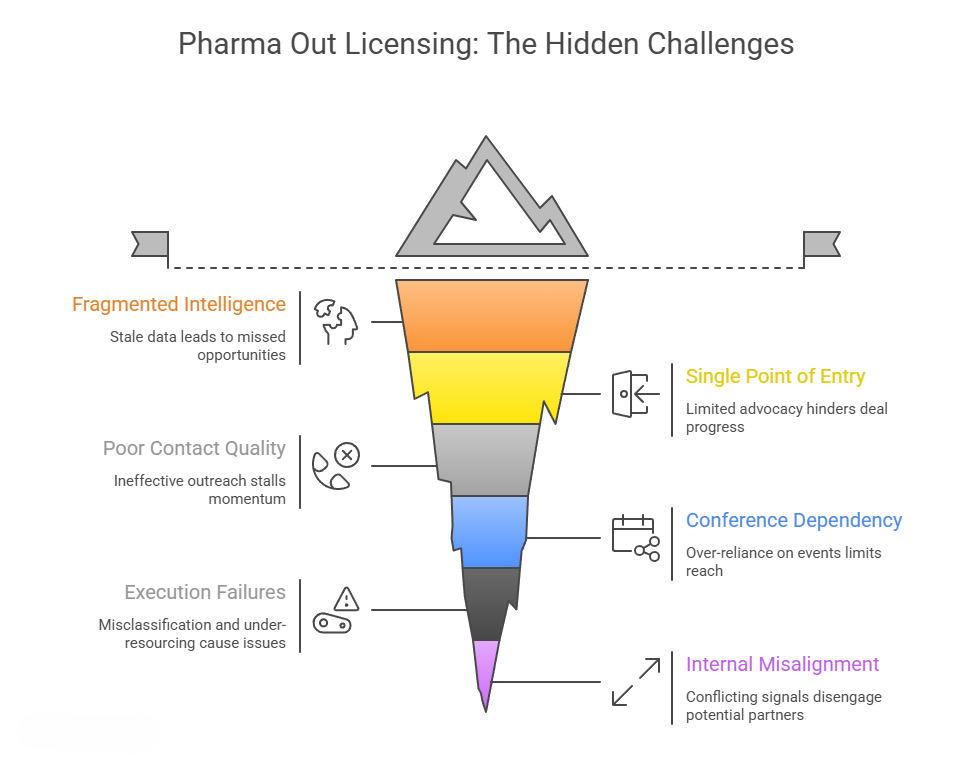
Out Licensing as a Strategic Instrument
Before diagnosing failure modes, it is worth anchoring out licensing within the broader corporate strategy framework. Out licensing is an inorganic growth strategy — it sits between pure in-house development and outright asset divestiture on the strategic spectrum. Its enduring appeal lies in risk-reward sharing: the licensor retains upside participation while offloading development capital and execution risk to a partner. For many biotech and specialty pharma companies, a well-structured licensing deal is not merely a revenue event — it is a capital-efficient mechanism for progressing multiple assets simultaneously.
The risk-adjusted net present value (rNPV) of an asset is directly tied to deal timing. Every month of delay has a measurable financial cost, which is why accurate valuation and NPV analysis should underpin the out licensing strategy from day one — informing both asset positioning and the acceptability of deal terms. According to IQVIA’s biopharma dealmaking analysis, licensing activity remained stable at around $170 billion in deal value even as M&A slowed — underscoring the enduring role of out licensing as a primary vehicle for accessing and externalising innovation.
Yet despite this strategic importance, out licensing is routinely treated as a secondary function. This misclassification is the root cause of most execution failures that follow.
Fragmented Intelligence on the Partner Landscape
The biopharma partnering landscape is not static. Companies pivot their therapeutic focus, acquire new capabilities, and disclose strategic priorities through earnings calls and R&D days in ways that are rarely captured in standard CRM lists or conference attendee directories. Yet a striking number of out licensing campaigns are built on precisely this kind of stale intelligence.
The consequence is predictable: outreach is sent to companies whose strategic priorities have shifted, while genuinely receptive partners — mid-sized biotechs with newly declared adjacency interests, or specialty pharma companies diversifying into a relevant indication — are never approached. As we explored in our piece on biotech partnering beyond the usual suspects, the universe of qualified partners is almost always broader than intuition suggests.
The effective counter is to treat partner mapping as a live competitive intelligence function. This means monitoring pipeline developments across primary targets and adjacent therapy areas, triangulating against public disclosures from R&D events, and explicitly identifying companies whose deal appetite is signalled by recent activity rather than historical precedent.
Single Point of Entry
Partnering decisions are organisationally distributed. Scientific, commercial, legal, and clinical development teams all contribute to a licensee’s internal assessment. Yet many out licensing campaigns are operationally structured as single-contact efforts — directed at a business development lead who functions as a gatekeeper rather than a champion.
The failure mode is not accessing the gatekeeper — it is stopping there. Business development contacts receive a high volume of inbound enquiries and are structurally incentivised to filter rather than advocate. An asset that enters a licensee organisation through a single contact, without internal advocacy from scientific or commercial colleagues, is disproportionately likely to stall or be quietly deprioritised.
The corrective is a multipronged engagement strategy. Business development contacts remain the logical entry point, but outreach should be designed to activate internal advocates across commercial, clinical, legal, and search-and-evaluation teams. This is resource-intensive by design and is best deployed once an appropriate non-confidential teaser is prepared — the teaser does the advocacy work inside the organisation when direct access to every stakeholder is impractical.
Contact Quality and Channel Discipline
Even when the right company is identified and the right stakeholders mapped, poor contact access can stall momentum indefinitely. Generic inboxes — bd@company.com — are processed slowly if at all. Outdated directories generate bounce rates that corrode credibility. Passive networking at conferences, while valuable for relationship maintenance, is insufficient as a primary outreach mechanism.
The most effective out licensing campaigns treat contact quality as a distinct asset. Warm introductions, trusted intermediaries, and direct access to named stakeholders materially improve response rates. As noted in our complete guide to pharma and biotech out licensing and partnering, building and maintaining this contact network is resource-intensive work, which makes it a strong candidate for delegation to external groups with established biopharma relationships, where the cost is offset by speed and conversion improvements.
The Conference Dependency Problem
Industry conferences — BIO International, BIO-Europe, ASCO, ESMO — are valuable forums. They concentrate decision-makers in one place and create efficient conditions for initial dialogue. The problem arises when they become the primary, or exclusive, mechanism for out licensing outreach.
Relying on conferences alone introduces structural limitations: deal timelines become anchored to event calendars, dormant contacts are never reactivated between conferences, and the concentrated competition for attention at major events reduces differentiation. It is also, as one practitioner aptly describes it, akin to speed dating — structurally unsuited to building the sustained dialogue that complex licensing negotiations require.
The appropriate use of conferences is additive rather than foundational. A year-round, proactive outreach rhythm — with conferences serving as acceleration points rather than starting guns — materially improves pipeline development and deal conversion. This aligns with the broader principle that accelerating pharma and biotech out licensing deals requires consistent, structured effort throughout the year, not periodic bursts.
Execution and Resourcing Failures
Three interconnected execution failures account for a disproportionate share of underperforming out licensing processes.
The first is structural misclassification: out licensing treated as an ancillary activity rather than a core business function. When responsibility is dispersed across individuals carrying other mandates, the result is not merely slower execution — it is the gradual erosion of rNPV as the asset ages without a deal.
The second is resourcing deficiency. Many organisations deploy a fractional business development officer as the primary vehicle for out licensing. The issue is not the individual’s experience — it is the absence of supporting infrastructure: intelligence, contact networks, deal-preparation resources, and project management bandwidth. Out licensing leadership is one component of an effective out licensing engine, not a substitute for it.
The third is preparation quality. Licensing packs — teasers, information memoranda, data room materials — are frequently assembled from generic internal presentations not designed for an external deal audience. Materials that lack a coherent investment story, that lead with scientific complexity ahead of commercial framing, or that assert best-in-class positioning without evidential support, create friction at precisely the moment a licensee’s interest is being formed. This is well documented in our analysis of eight critical barriers that undermine biotech out licensing deals.
Preparation is also a credibility exercise. An important but underappreciated dimension is the role of scientific due diligence: licensees conduct rigorous technical assessment, and materials that cannot withstand that scrutiny create deal-threatening friction during due diligence. Materials prepared with the involvement of external advisors — who bring objectivity, commercial framing, and an understanding of what licensees are actually looking for — consistently outperform internally produced equivalents. Our licensing and partnering advisory service is specifically structured to address these preparation gaps.
Internal Alignment: An Underappreciated Prerequisite
A final category of failure emerges not from the out licensing process itself, but from within the licensor’s own organisation. Misalignment between founders, investors, and internal teams — on deal parameters, geographic priorities, acceptable terms, or the interpretation of clinical data — creates a particularly damaging failure mode. A licensee that receives conflicting signals from different members of a licensor’s team will disengage, and rarely announces its reasons for doing so.
Internal alignment is not a process that can be completed on the fly during live negotiations. It requires explicit pre-process work: agreement on deal structure parameters and fallback positions, clarity on which territories are strategic versus opportunistic, and a shared understanding of how external feedback from potential licensees will be processed and acted upon. Comparative evaluation frameworks applied during the preparation phase can stress-test positioning assumptions before they are tested in live dialogue.
Market feedback from potential licensees is a valuable strategic input — organisations that treat it as such refine their positioning iteratively rather than persisting with a fixed narrative that is not resonating. This feedback loop also has competitive intelligence value: it surfaces how the asset is perceived relative to competing programmes, informing both the licensing narrative and broader competitive benchmarking exercises.
The Value Destruction Hidden in Plain Sight
Out licensing underperformance rarely announces itself as a crisis. Deals that fail to close are attributed to partner fit, market conditions, or asset characteristics. Timelines that extend to two years instead of twelve months are treated as inherent to the process. In both cases, the contribution of avoidable execution failures goes unaccounted.
The seven failure modes described here — fragmented partner intelligence, single-point-of-entry outreach, poor contact quality, conference dependency, structural misclassification, underpowered execution, and internal misalignment — are each individually correctable. Addressed collectively, they convert out licensing from an unpredictable process into a managed, high-velocity deal function. The scale of the market makes execution quality consequential: J.P. Morgan’s biopharma deal report shows that biopharma licensing deals reached $250 billion across 516 transactions — a market of this size rewards companies that run disciplined out licensing programmes and penalises those that do not.
For biotech and specialty pharma companies where a single licensing transaction can determine the trajectory of the organisation, that conversion is not a marginal improvement. It is a material driver of value. A useful reference point when assessing the financial stakes is our guide to pharma and biotech valuation, which provides the NPV and rNPV frameworks that quantify precisely what is at risk when out licensing execution falls short.
BiopharmaVantage provides specialist licensing and partnering, due diligence and competitive intelligence services to pharmaceutical and biotechnology companies. To discuss how we can support your out licensing programme, contact us.
