Agentic AI Competitive Intelligence in Pharma: The Strategic Imperative
Competitive intelligence has long served as a foundational pillar for strategic decision-making in the pharmaceutical industry. Yet today, the CI function stands at a critical inflection point. The evolution the industry is witnessing is not a distant transformation on the horizon – it is an immediate, fundamental shift in competitive intelligence capabilities. We are moving from manual, periodic competitor tracking to autonomous, continuous agentic systems that operate with minimal human intervention.
For CI leaders navigating this transition, adopting an agentic AI framework is no longer a discretionary AI/technology experiment. It has become a strategic imperative – one that directly impacts value via efficient performance and ultimately NPV maximization. Organizations that delay this transition risk falling behind competitors who are already leveraging AI-driven intelligence to make faster, more informed decisions.
This article examines the shift from generative to agentic AI in pharmaceutical competitive intelligence, explores the operational and financial benefits, addresses critical implementation challenges, and provides a practical roadmap for CI teams.
 From Generative AI to Agentic AI: Understanding the Shift
From Generative AI to Agentic AI: Understanding the Shift
Most pharmaceutical CI teams have already integrated the first wave of artificial intelligence into their workflows. This initial phase, often termed “Generative AI,” primarily utilizes Large Language Models (LLMs) to summarize clinical trial abstracts, scan news feeds, or generate report summaries. While this represents meaningful progress from purely manual processes, generative AI remains fundamentally reactive. It requires continuous human prompting, constant oversight, and significant manual intervention to extract actionable intelligence.
The next (rather current) frontier in competitive intelligence is Agentic AI – a paradigm shift that moves beyond simple text synthesis to autonomous reasoning and decision-making. Unlike standard AI tools that wait passively for user commands, agentic AI deploys specialized “agents” designed to execute complete CI workflows independently. These agents don’t simply answer questions when asked; they continuously monitor designated information sources, apply reasoning frameworks to identify material changes, and proactively alert teams when strategically significant events occur.
Consider a practical example: Rather than an analyst manually checking ClinicalTrials.gov each week for protocol amendments, an agentic system deploys a specialized clinical trials monitoring agent. This agent continuously scans the registry, applies domain-specific reasoning to interpret changes – such as recognizing that a Phase II primary endpoint modification likely signals localized recruitment challenges and will probably delay regulatory filing timelines – and alerts the team immediately with contextualized implications. The agent operates as a tireless, reasoning monitor that never sleeps, never takes vacation, and never misses a critical update. This shift from reactive querying to proactive monitoring represents a fundamental transformation in how competitive intelligence operates within pharmaceutical organizations.
The Multi-Agent Orchestration Framework
Forward-thinking CI teams are now adopting what industry experts call the Multi-Agent Orchestration framework. In this architectural approach, multiple specialist agents work in coordination to monitor the competitive landscape across several critical intelligence pillars such as:
Clinical Intelligence Agents continuously track inter alia:
- Trial registry updates and protocol amendments across global databases
- Site activation metrics and recruitment velocity indicators
- Investigator changes and clinical endpoint modifications
- Regulatory submission timelines and approval probability shifts
Commercial Intelligence Agents monitor amongst others:
- Pricing dynamics and reimbursement landscape changes
- Market access updates and formulary positioning shifts
- Regulatory approvals and label expansion opportunities
- Competitive promotional activity and messaging evolution
- Social media sentiments
Deal-Making Agents scan for events and indicators such as:
- Early signals of licensing interest and partnership intent
- M&A “noise” including executive movements and strategic announcements
- Patent activity, including extensions, challenges, and new filings
- Equity, debt, convertible raises, VC investments, and in-house investments
This orchestrated approach enables what is known as the “Search-to-Synthesis” evolution. Rather than simply describing historical events, the multi-agent system synthesizes cross-functional data points to generate Prescriptive CI – intelligence that helps predict what competitors are likely to do next.
For example, the system might correlate three seemingly unrelated signals: (1) a patent term extension filing, (2) a localized manufacturing delay at a contract manufacturer, and (3) delayed site activations in a specific geography. By connecting these dots, the agentic system can forecast a competitor’s supply-chain vulnerability window – providing your commercial team with a precise opportunity to gain share in that market before the competitor recovers. This level of sophisticated, cross-functional intelligence synthesis was previously possible only through extensive manual analysis by senior CI strategists. Agentic AI democratizes this capability, making it scalable and continuous.
Core Operational Benefits of Agentic CI
Deploying an agentic AI framework delivers multiple immediate, measurable operational advantages that directly impact CI team effectiveness:
1. Accelerated Speed to Insight
Time is currency in competitive intelligence. The faster an organization can move from raw data to strategic decision, the greater is the competitive advantage. Agentic AI systems dramatically compress the “Data-to-Decision” timeline by automating the most time-intensive phases of the intelligence cycle.
Industry benchmarks suggest that organizations implementing agentic CI frameworks reduce their intelligence cycle time by 60–70%. What previously required a week of manual monitoring, data collection, and preliminary analysis can now be completed in hours – or even minutes for high-priority intelligence alerts.
This acceleration matters enormously in fast-moving competitive scenarios. Consider a competitor’s unexpected clinical trial failure or a sudden regulatory setback. In traditional CI workflows, one might learn about this event days after it occurs, once the CI team processes routine monitoring reports. With agentic AI, one can receive an alert within hours, complete with preliminary strategic implications – giving the organization a critical head start in making optimal decisions.
2. Strategic Resource Optimization
CI Workflow Redesign: One of the most significant but often overlooked benefits of agentic AI is how it fundamentally reshapes team capacity allocation. By automating the labor-intensive “grunt work” of secondary research – scanning databases, monitoring news feeds, tracking registry updates – agentic systems free high-value internal CI team to focus high value adding activities such as:
- Intelligence Output Validation: Reviewing AI outputs and tweaking them to remove false inferences or flawed conclusions, ensuring the “machine signal” is grounded in reality.
- Communications: Communicating effectively across the organization to ensure intelligence is translated into actions for appropriate stakeholders.
- Intelligence Voids: Identifying critical intelligence gaps that remains and how to bridge them.
CI Resource Rebalancing: Instead of dedicating 70% of team capacity to routine monitoring and 30% to high-value activities, organizations flip this ratio.
3. Continuous 24/7 Coverage
Human-led CI operations inevitably contain coverage gaps. Internal CI teams and external CI vendors work within business hours constrained by geography and time zones. These inherently create intelligence blind spots – periods when critical competitor moves might go unnoticed for days or weeks despite all efforts.
Agentic AI systems eliminate these gaps entirely. They provide truly continuous 24/7 monitoring, scanning designated information sources every hour of every day without interruption. A competitor’s late-Friday regulatory filing, a weekend clinical trial update, or a holiday-timed partnership announcement – all are captured and analyzed in real-time, regardless of when they occur.
This “always-on” capability is particularly valuable for global pharmaceutical companies monitoring competitors across multiple time zones and therapeutic areas. The agentic system never sleeps, ensuring that no material competitive events and developments goes undetected, regardless of timing.
The Business Case for Agentic AI in Pharma
The Value Added
The business case for agentic CI is compelling, but pharmaceutical CI teams ultimately require a clear financial justification for investments. The case for internal AI-driven competitive intelligence is fundamentally a P&L and NPV discussion, not merely an operational efficiency argument.
According to McKinsey research, for the pharmaceutical sector, agentic AI is projected to lift growth by 5.0 to 13.0 percentage points and increase EBITDA by 3.4 to 5.4 percentage points over the next three to five years. Half of this value stems from revenue acceleration through better asset acquisition and faster time-to-market, while the other half is driven by non-linear efficiencies in R&D, manufacturing, and administration. While the data for CI is obviously not available yet, but since CI assists in several of those activities, a significant contribution can be expected.
The more nuanced ROI calculation centers on the NPV of accelerated decision-making. In pharmaceutical markets, timing precision directly impacts portfolio value.
Consider these scenarios:
- Identifying a competitor’s pipeline vulnerability three months earlier allows you to accelerate your own asset’s development timeline to capture first-mover advantage
- Detecting early partnership signals enables you to engage a potential target before competitive soliciting of partners escalates
- Recognizing a competitor’s manufacturing constraint creates a temporary window to gain market share that compounds over years
- Spotting clinical trial design weaknesses allows you to differentiate your own program’s endpoints before enrollment begins
Each of these timing advantages carries substantial NPV implications, often measured in tens or hundreds of millions of dollars for major pharmaceutical assets. The traditional CI model – with its inherent delays from manual monitoring and periodic reporting – simply cannot deliver this level of temporal precision.
The Cost Structure
Furthermore, the cost structure of agentic AI creates favorable unit economics compared to traditional vendor relationships:
- Traditional Linear Scaling: External CI vendors typically charge fees that scale linearly with scope – more therapeutic areas or monitoring programs monitored equals proportionally higher costs.
- Internal Agentic Economics: An internal agentic system exhibits dramatically different economics. Once the infrastructure is established, monitoring 100 indications incurs little incremental cost.
This drives substantial cost savings by creating economies of scale as the organization’s intelligence requirements expand.
The Mandate for Internalization of Pharma CI
Despite the clear advantages of AI-driven competitive intelligence, many pharmaceutical companies continue to outsource the bulk of their secondary CI work to external vendors. However, a decisive industry trend is emerging toward internalizing the routine monitoring programs – and this shift is driven by a few compelling needs:
1. The Proprietary Data Moat
External CI vendors, regardless of their sophistication, can only access and analyze publicly available information. They cannot – and should not – have access to your organization’s proprietary “dark data”: internal trial results, confidential RWE datasets, failed development programs, strategic planning documents, and competitive insights gathered through primary research.
An internal agentic AI system changes this dynamic entirely. By safely combining public competitive signals with your private intelligence, you create a customized analytical lens that no external party can replicate. The system can correlate a competitor’s public clinical trial results with your own confidential insights about similar programs, generating strategic perspectives impossible to obtain otherwise.
This proprietary data integration creates a genuine competitive moat – a source of intelligence advantage that compounds over time as your internal knowledge base deepens.
2. Strategic Intent Protection
Every query you submit to an external CI platform reveals something about your strategic priorities. Asking a vendor to deep-dive on a specific competitor, therapeutic area, or partnership scenario broadcasts your interests to a third party which potentially could propagate.
Internalizing competitive intelligence queries protects strategic privacy. Your agentic system operates entirely within your secure infrastructure, ensuring that your competitive focus areas, concern points, and strategic explorations remain confidential, and this intelligence security matters enormously.
3. Non-Linear Economics and Permanent Asset Building
Traditional CI vendor relationships operate on an OpEx model. You pay recurring fees – typically structured via annual monitoring programs. These costs scale roughly linearly with your intelligence requirements, creating budget constraints that force prioritization and inevitably leave intelligence gaps.
Internalized agentic AI fundamentally changes this economic equation. The investment shifts from recurring OpEx to upfront CapEx – building a permanent intelligence asset rather than procuring temporary services. Once your internal system is operational, expanding coverage from 10 therapeutic areas to 50, or from 20 competitors to 100, or from 10 indications to 50 requires minimal incremental cost.
This non-linear economic model allows pharmaceutical companies to achieve comprehensive competitive coverage that would be prohibitively expensive through traditional CI vendor relationships. You’re building institutional capability that becomes more valuable over time, rather than perpetually paying to CI vendors for repeated work. These factors dramatically increase the ROI of CI and accruing NPV in pharma organizations.
Critical Implementation Challenges
CI leaders must approach agentic AI adoption with clear-eyed pragmatism about genuine implementation challenges. Success requires acknowledging and actively managing several critical risks:
The Hallucination Problem
AI systems, including advanced LLMs, can generate factually incorrect statements with complete confidence – a phenomenon known as “hallucination.” For competitive intelligence, where strategic decisions depend on factual accuracy, this presents an unacceptable risk.
The solution is mandatory Human-in-the-Loop (HITL) verification for all high-stakes intelligence. Agentic systems should flag and prioritize insights, but internal CI team must validate critical findings before they communicate to relevant stakeholders. Think of AI as dramatically improving signal detection – but human expertise remains essential for signal verification.
Data Integration Friction
Agentic AI requires access to a unified “Data Fabric” – integrated information infrastructure where the system can seamlessly query across clinical, commercial, regulatory, and proprietary datasets. If your R&D systems remain isolated from commercial databases, and both are disconnected from external monitoring feeds, the agentic system cannot deliver its full analytical potential.
Many pharmaceutical companies discover that their greatest obstacle to effective AI implementation isn’t the AI technology itself – it’s the fragmented state of their underlying data architecture. Successful agentic CI deployment often requires preliminary investments in data integration and governance.
The Blackbox Reasoning Risk
There’s a dangerous temptation to treat autonomous AI insights as infallible. When a sophisticated system generates a strategic recommendation, organizational dynamics can discourage questioning the machine’s logic. This “blackbox” mentality – accepting AI outputs without critical evaluation – represents a fundamental risk.
Effective implementation requires maintaining analytical skepticism. Every significant AI-generated insight should be reviewed, its reasoning chain should be audited, and its conclusions should be stress-tested through human expertise and alternative analytical frameworks. The goal is human-AI collaboration, not human abdication to algorithmic authority.
The Management Model Transformation
Deploying an agentic CI system with 50+ specialized agents working continuously doesn’t eliminate CI team workload – it fundamentally transforms it. CI managers shift from executing research tasks to directing and prioritizing a digital workforce that generates constant alerts, insights, and intelligence flags.
This requires new management capabilities: deciding which agent-generated signals warrant human investigation, calibrating agent sensitivity to reduce false positives, and orchestrating agent priorities as strategic focuses shift. In many ways, managing an agentic system is more demanding than traditional human based CI – essentially, one is managing several digital CI analysts who are constantly producing output, and you are the sole project manager.
Agentic AI Competitive Intelligence in Pharma – Implementation Roadmap
Organizations ready to implement agentic competitive intelligence should follow a pragmatic, staged approach:
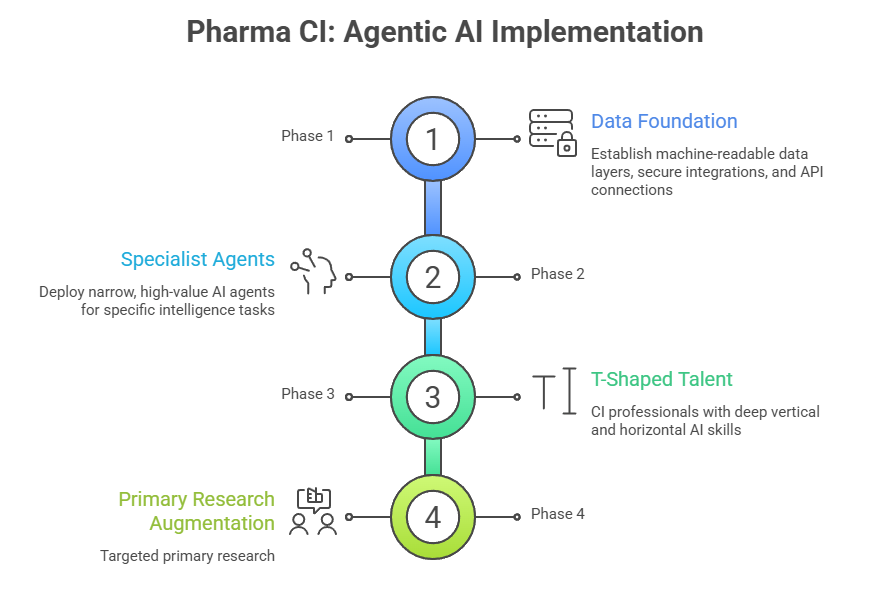
Phase 1: Establish the Data Foundation
Before deploying sophisticated AI agents, ensure your data infrastructure can support them. This means:
- Creating machine-readable data layers for internal information
- Establishing secure integration pathways between AI systems and proprietary databases
- Implementing governance frameworks that balance access with security
- Building API connections to critical external data sources
Phase 2: Deploy Narrow, High-Value Specialist Agents
Resist the temptation to build a broad, general-purpose system immediately. Instead, identify specific high-value, well-defined intelligence tasks in conjunction with CI leads where automation delivers clear ROI. Examples include:
- An “Oncology Patent Watcher” monitoring all patent filings in specific tumor types
- A “Clinical Endpoint Tracker” flagging protocol modifications across Phase III trials
- A “Pricing Intelligence Agent” monitoring reimbursement changes in key markets
Start narrow, prove value, then expand scope systematically.
Phase 3: Develop “T-Shaped” CI Talent
Agentic AI changes the required skill profile for competitive intelligence professionals. The most effective teams develop “T-shaped” capabilities:
- Deep vertical expertise in pharmaceutical strategy, therapeutic areas, and competitive dynamics (the vertical bar of the “T”)
- Horizontal skills in AI system management, data interpretation, and technology fluency (the horizontal bar of the “T”)
Invest in training programs that help experienced CI analysts transition from being “intelligence collectors” to “strategic pilots” who direct, verify, and synthesize AI-generated intelligence.
Phase 4: Augment with Strategic Primary Research
Critically, agentic AI does not eliminate – and in fact augments – the need for primary research (HUMINT). AI excels at monitoring what competitors publicly disclose. Primary research accesses what they have not disclosed: strategic intent revealed through expert conversations, relationship signals, cultural dynamics, and tacit decision-making.
The most effective competitive intelligence programs use AI to achieve comprehensive secondary coverage at minimal cost, then redirect saved resources toward targeted primary intelligence projects using external CI vendors that adds genuine value.
Conclusion: The Path Forward
The pharmaceutical competitive intelligence is accelerating toward a trend where most pharma companies own their internal intelligence infrastructure. By transitioning from externalized vendor engagements for routine secondary tracking toward internal, agentic AI frameworks, pharma CI teams can deliver the precision, speed, and comprehensive coverage.
This transformation is not about technology for technology’s sake. It’s about fundamentally improving the efficiency and efficacy of competitive intelligence enabling faster decisions, better resource allocation, and ultimately superior strategic outcomes. The ROI manifests not merely in cost savings, but in the compounding NPV benefits of consistently making better-informed decisions.
Organizations that successfully navigate this transition will establish a sustained competitive intelligence advantage. Those that delay face the prospect of falling permanently behind competitors who have already implements AI-driven competitive intelligence for decision-making. The question is no longer whether to adopt agentic competitive intelligence, but how quickly your organization can execute the transition.
BiopharmaVantage specializes in providing premium quality competitive intelligence services and wider decision-making services to pharma, biotech and diagnostics companies. If you would like to explore how we can assist you, please contact us.

 From Generative AI to Agentic AI: Understanding the Shift
From Generative AI to Agentic AI: Understanding the Shift